Greater access to high-quality rehabilitation treatments after stroke has the potential to improve movement and reduce disability for millions of people around the world. The National Institutes of Health recently awarded a large-scale five year grant to support a multi-site clinical trial investigating an intensive, home-based telerehabilitation treatment in persons post-stroke. The TR-2 trial was funded jointly by the National Institute of Neurological Disorders and Stroke (NINDS) and the Eunice Kennedy Shriver National Institute of Child Health and Human Development (NICHD).
The trial will be co-led by Steven C. Cramer, MD, Professor of Neurology, UCLA School of Medicine, and Medical Director of Research at California Rehabilitation Hospital, and Dylan Edwards, PhD, the Nancy Wachtel Shrier Director of Jefferson Moss Rehabilitation Research Institute and Professor of Rehabilitation Medicine at Thomas Jefferson University in Philadelphia. Both investigators have extensive experience with clinical trials involving rehabilitation robotics, technology and digital gaming and have spent substantial portions of their careers studying recovery of post-stroke motor function. The approach used in the TR-2 trial is built on current theory of motor recovery treatment combined with contemporary technology to improve access to stroke rehabilitation.
After years of study through smaller clinical trials, this new randomized, controlled trial aims to definitively establish the value of high-dose remote rehabilitation, an approach originally developed by Dr. Cramer and colleagues in his laboratory. Dr. Cramer recognized early on that applying a telehealth approach to stroke rehabilitation had tremendous potential for increasing the dose of therapy that patients could receive while overcoming some of the many barriers that limit the amount of therapy delivered through traditional clinic visits, including limited access, difficulty traveling, and low motivation.
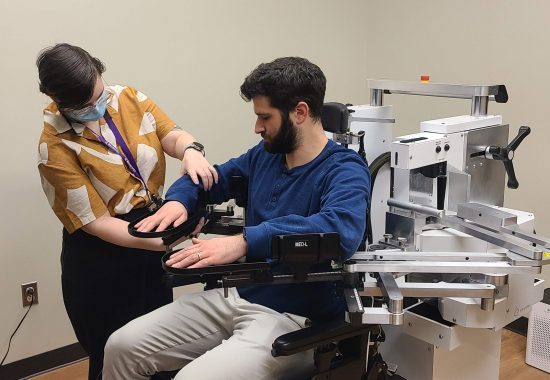
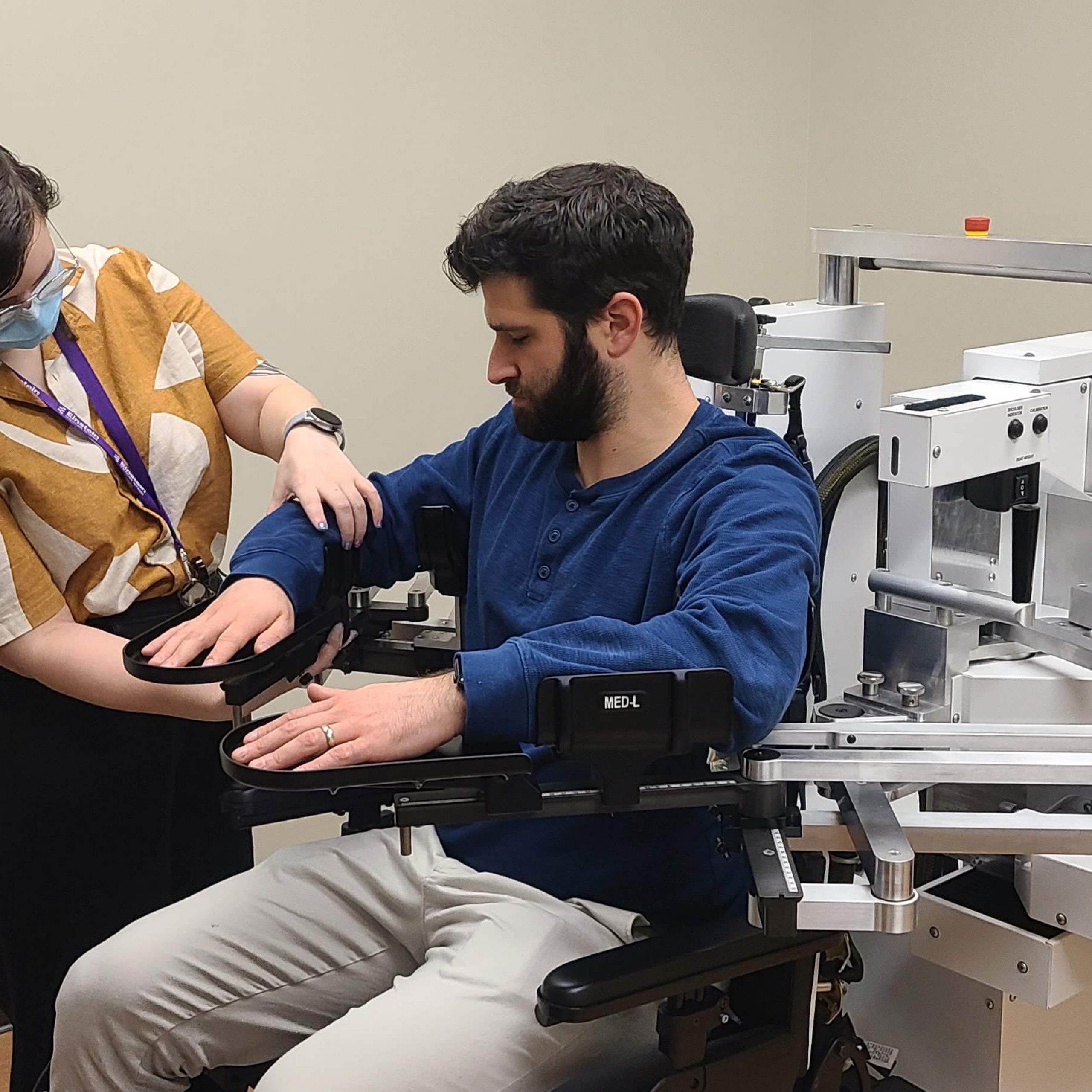
“Using telerehabilitation to facilitate the recovery of motor function after stroke made sense. To get this approach into the clinic, we first had the important task of working out the logistics and designing an intervention that was safe, effective, and — importantly — something that people actually wanted to do,” explained Dr. Cramer. Dr. Edwards has been collaborating with Dr. Cramer in this research area for more than a decade, and the positive findings from Dr. Cramer’s preliminary trial published in 2017 were encouraging. They have worked together to continue to refine the intervention, which consists of motor therapy targeting arm function through engaging functional games and exercise videos, as well as stroke education and regular assessments.
Working with a team of exceptional researchers from eleven different sites across the United States, their results from their earlier Phase II trial demonstrated that activity-based training produced substantial gains in arm motor function, regardless of whether the training was provided via home-based telerehabilitation or traditional rehabilitation in a clinical setting (non-inferiority trial). In a recent dual-site study, they also determined that this intensive telerehabilitation approach is feasible, safe, and potentially effective to use for individuals early after a stroke while they are still in an inpatient rehabilitation facility and transition to home with it.
The TR-2 trial builds on collective previous findings of feasibility, safety, and efficacy, but importantly, this is the first phase II progression to a phase III pivotal superiority trial funded by the NIH (StrokeNet). It will do this by directly comparing intensive, home-based rehabilitation therapy to usual clinical care to determine whether the telerehabilitation approach is more effective. This study brings together a multi-disciplinary team of co-investigators with expertise across stroke rehabilitation, neuroimaging, actigraphy, health economics, biostatistics, and clinical trials, as well as experienced research therapists and clinical coordinators at the lead sites.
Beyond examining the effectiveness of adding the intensive, home-based telerehabilitation treatment versus usual care in a larger sample of more than 200 participants across many sites, their newly funded study will evaluate a neuroimaging-based measure of stroke injury that can be used to predict how an individual may benefit from this intensive rehabilitation therapy. This measure will specifically examine the extent of damage to the descending pathways involved in voluntary movement (corticospinal tract), a measure that has been shown to predict arm motor recovery after stroke. They hypothesize that the beneficial effects of their targeted motor therapy depend on having an intact corticospinal tract, and the extent of corticospinal preservation should be related to the degree of benefits this therapy can provide over usual care.
“Determining which patients will respond best to which therapies is a challenge both in everyday clinical practice and in clinical trials because of the large heterogeneity of stroke. Identifying a measure that can successfully predict who will benefit from a particular therapy is critical for optimizing outcomes and utilization for patients who may have a limited amount of time and other resources to dedicate to rehabilitation,” Dr. Edwards remarked.
In addition to causing widespread disability, stroke contributes to significant economic burden, both for individuals and the broader healthcare system. Telerehabilitation may be able to serve as a practical and cost-effective approach for delivering care that can improve function and reduce disability. As part of their new grant, the interdisciplinary research team will also examine the health economic impact of home-based telerehabilitation in patients with recent stroke, directly comparing it to usual care in which rehabilitation therapy is generally provided at very low doses.
Drs. Cramer and Edwards are looking forward to identifying the sites that will participate in the newly initiated TR-2 trial, and they anticipate participant enrollment will begin next year. By dramatically improving access to rehabilitation services and increasing the amount of therapy that individuals with stroke receive, the findings of this study have the potential to change clinical care for stroke rehabilitation worldwide.